Gaoni YMR. Isolation, structure, and partial synthesis of an active constituent of hashish. J Am Chem Soc. 1964;86:1646.
ElSohly, MA, Gul, W, Chapter 1: constituents of Cannabis sativa, in, R Pertwee, Editor. Handbook of Cannabis. Oxford University Press; 2014. pp. 3-22.
Rock EM, Parker LA. Constituents of Cannabis sativa. Adv Exp Med Biol. 2021;1264:1–13.
Andersen SL. Trajectories of brain development: point of vulnerability or window of opportunity? Neurosci Biobehav Rev. 2003;27:3–18.
Casey BJ, Giedd JN, Thomas KM. Structural and functional brain development and its relation to cognitive development. Biol Psychol. 2000;54:241–57.
Chambers RA, Taylor JR, Potenza MN. Developmental neurocircuitry of motivation in adolescence: a critical period of addiction vulnerability. Am J Psychiatry. 2003;160:1041–52.
Spear LP. The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev. 2000;24:417–63.
Crews F, He J, Hodge C. Adolescent cortical development: a critical period of vulnerability for addiction. Pharmacol Biochem Behav. 2007;86:189–99.
Arseneault L, Cannon M, Poulton R, Murray R, Caspi A, Moffitt TE. Cannabis use in adolescence and risk for adult psychosis: longitudinal prospective study. Bmj. 2002;325:1212–3.
Caspi A, Moffitt TE, Cannon M, McClay J, Murray R, Harrington H, et al. Moderation of the effect of adolescent-onset Cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: longitudinal evidence of a gene X environment interaction. Biol Psychiatry. 2005;57:1117–27.
Ferland JN, Ellis RJ, Betts G, Silveira MM, de Firmino JB, Winstanley CA, et al. Long-Term Outcomes of adolescent THC exposure on translational cognitive measures in adulthood in an animal model and computational assessment of human data. JAMA Psychiatry. 2023;80:66–76.
Harvey MA, Sellman JD, Porter RJ, Frampton CM. The relationship between non-acute adolescent Cannabis use and cognition. Drug Alcohol Rev. 2007;26:309–19.
Sewell RA, Ranganathan M, D’Souza DC. Cannabinoids and psychosis. Int Rev Psychiatry. 2009;21:152–62.
Zammit S, Allebeck P, Andreasson S, Lundberg I, Lewis G. Self reported Cannabis use as a risk factor for schizophrenia in Swedish conscripts of 1969: historical cohort study. Bmj. 2002;325:1199.
Di Forti M, Quattrone D, Freeman TP, Tripoli G, Gayer-Anderson C, Quigley H, et al. The contribution of Cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): a multicentre case-control study. Lancet Psychiatry. 2019;6:427–36.
Murray RM, Englund A, Abi-Dargham A, Lewis DA, Di Forti M, Davies C, et al. Cannabis-associated psychosis: neural substrate and clinical impact. Neuropharmacology. 2017;124:89–104.
Moore TH, Zammit S, Lingford-Hughes A, Barnes TR, Jones PB, Burke M, et al. Cannabis use and risk of psychotic or affective mental health outcomes: a systematic review. Lancet. 2007;370:319–28.
Murray RM, Di Forti M. Cannabis and psychosis: what degree of proof do we require? Biol Psychiatry. 2016;79:514–5.
Caballero A, Tseng KY. Association of Cannabis use during adolescence, prefrontal CB1 receptor signaling, and schizophrenia. Front Pharmacol. 2012;3:101.
Hurd YL, Manzoni OJ, Pletnikov MV, Lee FS, Bhattacharyya S, Melis M. Cannabis and the developing brain: insights into its long-lasting effects. J Neurosci. 2019;39:8250–8.
Scheyer AF, Melis M, Trezza V, Manzoni OJJ. Consequences of perinatal Cannabis exposure. Trends Neurosci. 2019;42:871–84.
Molla HM, Tseng KY. Neural substrates underlying the negative impact of cannabinoid exposure during adolescence. Pharmacol Biochem Behav. 2020;195:172965.
Bara A, Ferland JN, Rompala G, Szutorisz H, Hurd YL. Cannabis and synaptic reprogramming of the developing brain. Nat Rev Neurosci. 2021;22:423–38.
Stringfield SJ, Torregrossa MM. Intravenous self-administration of delta-9-THC in adolescent rats produces long-lasting alterations in behavior and receptor protein expression. Psychopharmacology. 2021;238:305–19.
Peters KZ, Zlebnik NE, Cheer JF. Cannabis exposure during adolescence: a uniquely sensitive period for neurobiological effects. Int Rev Neurobiol. 2022;161:95–120.
Li Z, Mukherjee D, Duric B, Austin-Zimmerman I, Trotta G, Spinazzola E, et al. Systematic review and meta-analysis on the effects of chronic peri-adolescent cannabinoid exposure on schizophrenia-like behaviour in rodents. Mol Psychiatry. 2024;30:285–95.
Rubino T, Parolaro D. The impact of exposure to cannabinoids in adolescence: insights from animal models. Biol Psychiatry. 2016;79:578–85.
Bossong MG, Niesink RJ. Adolescent brain maturation, the endogenous cannabinoid system and the neurobiology of Cannabis-induced schizophrenia. Prog Neurobiol. 2010;92:370–85.
Ferland JN, Hurd YL. Deconstructing the neurobiology of Cannabis use disorder. Nat Neurosci. 2020;23:600–10.
Renard J, Rushlow WJ, Laviolette SR. Effects of adolescent THC exposure on the prefrontal GABAergic system: implications for schizophrenia-related psychopathology. Front Psychiatry. 2018;9:281.
Caballero A, Tseng KY. GABAergic function as a limiting factor for prefrontal maturation during adolescence. Trends Neurosci. 2016;39:441–8.
Colizzi M, McGuire P, Pertwee RG, Bhattacharyya S. Effect of Cannabis on glutamate signalling in the brain: a systematic review of human and animal evidence. Neurosci Biobehav Rev. 2016;64:359–81.
Devane WA, Dysarz FA 3rd, Johnson MR, Melvin LS, Howlett AC. Determination and characterization of a cannabinoid receptor in rat brain. Mol Pharmacol. 1988;34:605–13.
Mallet PE, Beninger RJ. The cannabinoid CB1 receptor antagonist SR141716A attenuates the memory impairment produced by delta9-tetrahydrocannabinol or anandamide. Psychopharmacology. 1998;140:11–19.
Raver SM, Keller A. Permanent suppression of cortical oscillations in mice after adolescent exposure to cannabinoids: receptor mechanisms. Neuropharmacology. 2014;86:161–73.
D’Souza DC, Perry E, MacDougall L, Ammerman Y, Cooper T, Wu YT, et al. The psychotomimetic effects of intravenous delta-9-tetrahydrocannabinol in healthy individuals: implications for psychosis. Neuropsychopharmacology. 2004;29:1558–72.
Englund A, Atakan Z, Kralj A, Tunstall N, Murray R, Morrison P. The effect of five day dosing with THCV on THC-induced cognitive, psychological and physiological effects in healthy male human volunteers: a placebo-controlled, double-blind, crossover pilot trial. J Psychopharmacol. 2016;30:140–51.
Horder J, Browning M, Di Simplicio M, Cowen PJ, Harmer CJ. Effects of 7 days of treatment with the cannabinoid type 1 receptor antagonist, rimonabant, on emotional processing. J Psychopharmacol. 2012;26:125–32.
Zuurman L, Roy C, Schoemaker RC, Amatsaleh A, Guimaeres L, Pinquier JL, et al. Inhibition of THC-induced effects on the central nervous system and heart rate by a novel CB1 receptor antagonist AVE1625. J Psychopharmacol. 2010;24:363–71.
Herkenham M, Lynn AB, Johnson MR, Melvin LS, de Costa BR, Rice KC. Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J Neurosci. 1991;11:563–83.
Herkenham M, Lynn AB, Little MD, Johnson MR, Melvin LS, de Costa BR, et al. Cannabinoid receptor localization in brain. Proc Natl Acad Sci USA. 1990;87:1932–6.
Lovinger DM. Presynaptic modulation by endocannabinoids. Handb Exp Pharmacol. 2008;184:435–77.
Freund TF, Katona I, Piomelli D. Role of endogenous cannabinoids in synaptic signaling. Physiol Rev. 2003;83:1017–66.
Regehr WG, Carey MR, Best AR. Activity-dependent regulation of synapses by retrograde messengers. Neuron. 2009;63:154–70.
Cass DK, Flores-Barrera E, Thomases DR, Vital WF, Caballero A, Tseng KY. CB1 cannabinoid receptor stimulation during adolescence impairs the maturation of GABA function in the adult rat prefrontal cortex. Mol Psychiatry. 2014;19:536–43.
Molla HM, Miguelez Fernandez AMM, Tseng KY. Late-adolescent onset of prefrontal endocannabinoid control of hippocampal and amygdalar inputs and its impact on trace-fear conditioning behavior. Neuropsychopharmacology. 2024;49:1417–24.
Choi K, Le T, McGuire J, Xing G, Zhang L, Li H, et al. Expression pattern of the cannabinoid receptor genes in the frontal cortex of mood disorder patients and mice selectively bred for high and low fear. J Psychiatr Res. 2012;46:882–9.
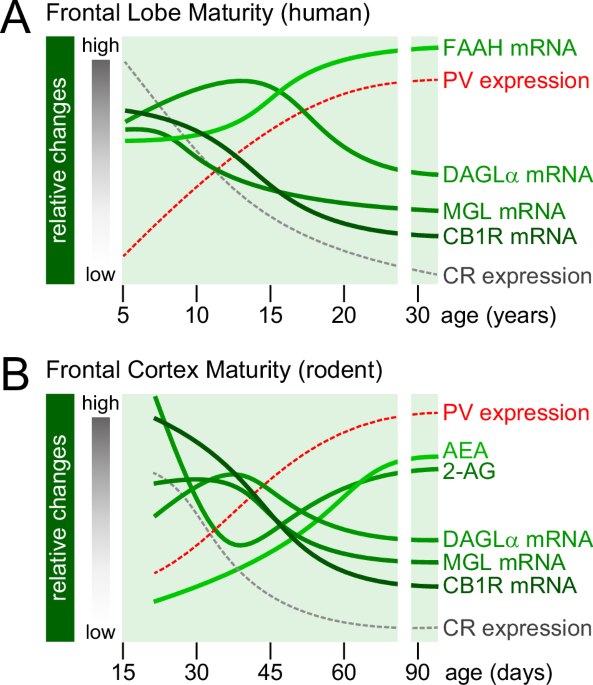
Long LE, Lind J, Webster M, Weickert CS. Developmental trajectory of the endocannabinoid system in human dorsolateral prefrontal cortex. BMC Neurosci. 2012;13:87.
Heng L, Beverley JA, Steiner H, Tseng KY. Differential developmental trajectories for CB1 cannabinoid receptor expression in limbic/associative and sensorimotor cortical areas. Synapse. 2011;65:278–86.
Chesworth R, Long LE, Weickert CS, Karl T. The endocannabinoid system across postnatal development in transmembrane domain neuregulin 1 mutant mice. Front Psychiatry. 2018;9:11.
Ellgren M, Artmann A, Tkalych O, Gupta A, Hansen HS, Hansen SH, et al. Dynamic changes of the endogenous cannabinoid and opioid mesocorticolimbic systems during adolescence: THC effects. Eur Neuropsychopharmacol. 2008;18:826–34.
Yang S, Tseng KY. Maturation of corticolimbic functional connectivity during sensitive periods of brain development. Curr Top Behav Neurosci. 2022;53:37–53.
Castillo PE, Younts TJ, Chavez AE, Hashimotodani Y. Endocannabinoid signaling and synaptic function. Neuron. 2012;76:70–81.
Kano M, Hashimoto K, Tabata T. Type-1 metabotropic glutamate receptor in cerebellar Purkinje cells: a key molecule responsible for long-term depression, endocannabinoid signalling and synapse elimination. Philos Trans R Soc Lond B Biol Sci. 2008;363:2173–86.
Van Waes V, Beverley JA, Siman H, Tseng KY, Steiner H. CB1 cannabinoid receptor expression in the striatum: association with corticostriatal circuits and developmental regulation. Front Pharmacol. 2012;3:21.
Andersen, SL, Sensitive periods of brain development and preventive interventions. 2021/08/14 ed. Current Topics in Behavioral Neurosciences. Vol. 53. 2022.
Berghuis P, Dobszay MB, Wang X, Spano S, Ledda F, Sousa KM, et al. Endocannabinoids regulate interneuron migration and morphogenesis by transactivating the TrkB receptor. Proc Natl Acad Sci USA. 2005;102:19115–20.
Berghuis P, Rajnicek AM, Morozov YM, Ross RA, Mulder J, Urban GM, et al. Hardwiring the brain: endocannabinoids shape neuronal connectivity. Science. 2007;316:1212–6.
Mulder J, Aguado T, Keimpema E, Barabas K, Ballester Rosado CJ, Nguyen L, et al. Endocannabinoid signaling controls pyramidal cell specification and long-range axon patterning. Proc Natl Acad Sci USA. 2008;105:8760–5.
Watson S, Chambers D, Hobbs C, Doherty P, Graham A. The endocannabinoid receptor, CB1, is required for normal axonal growth and fasciculation. Mol Cell Neurosci. 2008;38:89–97.
Frau R, Miczan V, Traccis F, Aroni S, Pongor CI, Saba P, et al. Prenatal THC exposure produces a hyperdopaminergic phenotype rescued by pregnenolone. Nat Neurosci. 2019;22:1975–85.
Rodriguez de Fonseca F, Hernandez ML, de Miguel R, Fernandez-Ruiz JJ, Ramos JA. Early changes in the development of dopaminergic neurotransmission after maternal exposure to cannabinoids. Pharmacol Biochem Behav. 1992;41:469–74.
Scheyer AF, Borsoi M, Wager-Miller J, Pelissier-Alicot AL, Murphy MN, Mackie K, et al. Cannabinoid exposure via lactation in rats disrupts perinatal programming of the gamma-aminobutyric acid trajectory and select early-life behaviors. Biol Psychiatry. 2020;87:666–77.
de Salas-Quiroga A, Diaz-Alonso J, Garcia-Rincon D, Remmers F, Vega D, Gomez-Canas M, et al. Prenatal exposure to cannabinoids evokes long-lasting functional alterations by targeting CB1 receptors on developing cortical neurons. Proc Natl Acad Sci USA. 2015;112:13693–8.
de Salas-Quiroga A, Garcia-Rincon D, Gomez-Dominguez D, Valero M, Simon-Sanchez S, Paraiso-Luna J, et al. Long-term hippocampal interneuronopathy drives sex-dimorphic spatial memory impairment induced by prenatal THC exposure. Neuropsychopharmacology. 2020;45:877–86.
Spano MS, Ellgren M, Wang X, Hurd YL. Prenatal Cannabis exposure increases heroin seeking with allostatic changes in limbic enkephalin systems in adulthood. Biol Psychiatry. 2007;61:554–63.
Tortoriello G, Morris CV, Alpar A, Fuzik J, Shirran SL, Calvigioni D, et al. Miswiring the brain: Delta9-tetrahydrocannabinol disrupts cortical development by inducing an SCG10/stathmin-2 degradation pathway. EMBO J. 2014;33:668–85.
Ryan KS, Karpf JA, Chan CN, Hagen OL, McFarland TJ, Urian JW, et al. Prenatal delta-9-tetrahydrocannabinol exposure alters fetal neurodevelopment in rhesus macaques. Sci Rep. 2024;14:5808.
Chen CY, O’Brien MS, Anthony JC. Who becomes Cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000–2001. Drug Alcohol Depend. 2005;79:11–22.
Martin GI. Marijuana: the effects on pregnancy, the fetus, and the newborn. J Perinatol. 2020;40:1470–6.
Baranger DAA. Evaluating evidence supporting an effect of prenatal Cannabis exposure on white matter integrity. Biol Psychiatry Glob Open Sci. 2024;4:101–2.
Best JR, Miller PH. A developmental perspective on executive function. Child Dev. 2010;81:1641–60.
Tseng KY, Chambers RA, Lipska BK. The neonatal ventral hippocampal lesion as a heuristic neurodevelopmental model of schizophrenia. Behav Brain Res. 2009;204:295–305.
Scheyer AF, Borsoi M, Pelissier-Alicot AL, Manzoni OJJ. Maternal exposure to the cannabinoid agonist WIN 55,12,2 during lactation induces lasting behavioral and synaptic alterations in the rat adult offspring of both sexes. eNeuro. 2020;7:ENEURO.0144-20.2020.
Scheyer AF, Borsoi M, Pelissier-Alicot AL, Manzoni OJJ. Perinatal THC exposure via lactation induces lasting alterations to social behavior and prefrontal cortex function in rats at adulthood. Neuropsychopharmacology. 2020;45:1826–33.
Lujan MA, Young-Morrison R, Aroni S, Katona I, Melis M, Cheer JF. Dynamic overrepresentation of accumbal cues in food- and opioid-seeking rats after prenatal THC exposure. Sci Adv. 2024;10:eadq5652.
Peterson CS, Baglot SL, Sallam NA, Mina S, Hill MN, Borgland SL. Oral pre- and early postnatal Cannabis exposure disinhibits ventral tegmental area dopamine neuron activity but does not influence cocaine preference in offspring in mice. J Neurosci Res. 2024;102:e25369.
Frau R, Melis M. Sex-specific susceptibility to psychotic-like states provoked by prenatal THC exposure: reversal by pregnenolone. J Neuroendocrinol. 2023;35:e13240.
DiNieri JA, Wang X, Szutorisz H, Spano SM, Kaur J, Casaccia P, et al. Maternal Cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol Psychiatry. 2011;70:763–9.
Lew SE, Tseng KY. Dopamine modulation of GABAergic function enables network stability and input selectivity for sustaining working memory in a computational model of the prefrontal cortex. Neuropsychopharmacology. 2014;39:3067–76.
Flores-Barrera E, Thomases DR, Heng LJ, Cass DK, Caballero A, Tseng KY. Late adolescent expression of GluN2B transmission in the prefrontal cortex is input-specific and requires postsynaptic protein kinase A and D1 dopamine receptor signaling. Biol Psychiatry. 2014;75:508–16.
Satterthwaite TD, Wolf DH, Erus G, Ruparel K, Elliott MA, Gennatas ED, et al. Functional maturation of the executive system during adolescence. J Neurosci. 2013;33:16249–61.
Gee DG, Humphreys KL, Flannery J, Goff B, Telzer EH, Shapiro M, et al. A developmental shift from positive to negative connectivity in human amygdala-prefrontal circuitry. J Neurosci. 2013;33:4584–93.
Swartz JR, Phan KL, Angstadt M, Klumpp H, Fitzgerald KD, Monk CS. Altered activation of the rostral anterior cingulate cortex in the context of emotional face distractors in children and adolescents with anxiety disorders. Depress Anxiety. 2014;31:870–9.
Paus T, Zijdenbos A, Worsley K, Collins DL, Blumenthal J, Giedd JN, et al. Structural maturation of neural pathways in children and adolescents: in vivo study. Science. 1999;283:1908–11.
Sowell ER, Thompson PM, Holmes CJ, Batth R, Jernigan TL, Toga AW. Localizing age-related changes in brain structure between childhood and adolescence using statistical parametric mapping. Neuroimage. 1999;9:587–97.
Floresco SB, Seamans JK, Phillips AG. Selective roles for hippocampal, prefrontal cortical, and ventral striatal circuits in radial-arm maze tasks with or without a delay. J Neurosci. 1997;17:1880–90.
Ishikawa A, Nakamura S. Convergence and interaction of hippocampal and amygdalar projections within the prefrontal cortex in the rat. J Neurosci. 2003;23:9987–95.
Tse MT, Piantadosi PT, Floresco SB. Prefrontal cortical gamma-aminobutyric acid transmission and cognitive function: drawing links to schizophrenia from preclinical research. Biol Psychiatry. 2015;77:929–39.
Calabro FJ, Murty VP, Jalbrzikowski M, Tervo-Clemmens B, Luna B. Development of hippocampal-prefrontal cortex interactions through adolescence. Cereb Cortex. 2020;30:1548–58.
Murty VP, Calabro F, Luna B. The role of experience in adolescent cognitive development: integration of executive, memory, and mesolimbic systems. Neurosci Biobehav Rev. 2016;70:46–58.
Larsen B, Cui Z, Adebimpe A, Pines A, Alexander-Bloch A, Bertolero M, et al. A developmental reduction of the excitation:inhibition ratio in association cortex during adolescence. Sci Adv. 2022;8:eabj8750.
Caballero A, Orozco A, Tseng KY. Developmental regulation of excitatory-inhibitory synaptic balance in the prefrontal cortex during adolescence. Semin Cell Dev Biol. 2021;118:60–63.
Caballero A, Flores-Barrera E, Thomases DR, Tseng KY. Downregulation of parvalbumin expression in the prefrontal cortex during adolescence causes enduring prefrontal disinhibition in adulthood. Neuropsychopharmacology. 2020;45:1527–35.
Cass DK, Thomases DR, Caballero A, Tseng KY. Developmental disruption of gamma-aminobutyric acid function in the medial prefrontal cortex by noncontingent cocaine exposure during early adolescence. Biol Psychiatry. 2013;74:490–501.
Flores-Barrera E, Thomases DR, Tseng KY. MK-801 exposure during adolescence elicits enduring disruption of prefrontal E-I balance and its control of fear extinction behavior. J Neurosci. 2020;40:4881–7.
Thomases DR, Cass DK, Meyer JD, Caballero A, Tseng KY. Early adolescent MK-801 exposure impairs the maturation of ventral hippocampal control of basolateral amygdala drive in the adult prefrontal cortex. J Neurosci. 2014;34:9059–66.
Thomases DR, Cass DK, Tseng KY. Periadolescent exposure to the NMDA receptor antagonist MK-801 impairs the functional maturation of local GABAergic circuits in the adult prefrontal cortex. J Neurosci. 2013;33:26–34.
Cuccurazzu B, Zamberletti E, Nazzaro C, Prini P, Trusel M, Grilli M, et al. Adult cellular neuroadaptations induced by adolescent THC exposure in female rats are rescued by enhancing anandamide signaling. Int J Neuropsychopharmacol. 2018;21:1014–24.
Rubino T, Prini P, Piscitelli F, Zamberletti E, Trusel M, Melis M, et al. Adolescent exposure to THC in female rats disrupts developmental changes in the prefrontal cortex. Neurobiol Dis. 2015;73:60–69.
Pickel VM, Bourie F, Chan J, Mackie K, Lane DA, Wang G. Chronic adolescent exposure to ∆9-tetrahydrocannabinol decreases NMDA current and extrasynaptic plasmalemmal density of NMDA GluN1 subunits in the prelimbic cortex of adult male mice. Neuropsychopharmacology. 2020;45:374–83.
Miguelez Fernandez AMM, Molla HM, Thomases DR, Tseng KY. Prefrontal alpha7nAChR signaling differentially modulates afferent drive and trace fear conditioning behavior in adolescent and adult rats. J Neurosci. 2021;41:1908–16.
Heng LJ, Markham JA, Hu XT, Tseng KY. Concurrent upregulation of postsynaptic L-type Ca(2+) channel function and protein kinase A signaling is required for the periadolescent facilitation of Ca(2+) plateau potentials and dopamine D1 receptor modulation in the prefrontal cortex. Neuropharmacology. 2011;60:953–62.
Tseng KY, O’Donnell P. Post-pubertal emergence of prefrontal cortical up states induced by D1-NMDA co-activation. Cereb Cortex. 2005;15:49–57.
De Felice M, Chen C, Rodriguez-Ruiz M, Szkudlarek HJ, Lam M, Sert S, et al. Adolescent delta-9-tetrahydrocannabinol exposure induces differential acute and long-term neuronal and molecular disturbances in dorsal vs. ventral hippocampal subregions. Neuropsychopharmacology. 2023;48:540–51.
De Gregorio D, Dean Conway J, Canul ML, Posa L, Bambico FR, Gobbi G. Effects of chronic exposure to low doses of delta9- tetrahydrocannabinol in adolescence and adulthood on serotonin/norepinephrine neurotransmission and emotional behaviors. Int J Neuropsychopharmacol. 2020;23:751–61.
Kruse LC, Cao JK, Viray K, Stella N, Clark JJ. Voluntary oral consumption of delta(9)-tetrahydrocannabinol by adolescent rats impairs reward-predictive cue behaviors in adulthood. Neuropsychopharmacology. 2019;44:1406–14.
Pistis M, Perra S, Pillolla G, Melis M, Muntoni AL, Gessa GL. Adolescent exposure to cannabinoids induces long-lasting changes in the response to drugs of abuse of rat midbrain dopamine neurons. Biol Psychiatry. 2004;56:86–94.
Scherma M, Dessi C, Muntoni AL, Lecca S, Satta V, Luchicchi A, et al. Adolescent delta(9)-tetrahydrocannabinol exposure alters WIN55,212-2 self-administration in adult rats. Neuropsychopharmacology. 2016;41:1416–26.
Cajiao-Manrique MDM, Casado-Anguera V, Garcia-Blanco A, Maldonado R, Martin-Garcia E. THC exposure during adolescence increases impulsivity-like behavior in adulthood in a WIN 55,212-2 self-administration mouse model. Front Psychiatry. 2023;14:1148993.
Zamberletti E, Beggiato S, Steardo L Jr., Prini P, Antonelli T, Ferraro L, et al. Alterations of prefrontal cortex GABAergic transmission in the complex psychotic-like phenotype induced by adolescent delta-9-tetrahydrocannabinol exposure in rats. Neurobiol Dis. 2014;63:35–47.
Izquierdo-Luengo C, Ten-Blanco M, Ponce-Renilla M, Perezzan R, Pereda-Perez I, Berrendero F. Adolescent exposure to the Spice/K2 cannabinoid JWH-018 impairs sensorimotor gating and alters cortical perineuronal nets in a sex-dependent manner. Transl Psychiatry. 2023;13:176.
Kirschmann EK, Pollock MW, Nagarajan V, Torregrossa MM. Effects of adolescent cannabinoid self-administration in rats on addiction-related behaviors and working memory. Neuropsychopharmacology. 2017;42:989–1000.
Quinn HR, Matsumoto I, Callaghan PD, Long LE, Arnold JC, Gunasekaran N, et al. Adolescent rats find repeated delta(9)-THC less aversive than adult rats but display greater residual cognitive deficits and changes in hippocampal protein expression following exposure. Neuropsychopharmacology. 2008;33:1113–26.
Renard J, Rosen LG, Loureiro M, De Oliveira C, Schmid S, Rushlow WJ, et al. Adolescent cannabinoid exposure induces a persistent sub-cortical hyper-dopaminergic state and associated molecular adaptations in the prefrontal cortex. Cereb Cortex. 2017;27:1297–310.
Miller ML, Chadwick B, Dickstein DL, Purushothaman I, Egervari G, Rahman T, et al. Adolescent exposure to delta(9)-tetrahydrocannabinol alters the transcriptional trajectory and dendritic architecture of prefrontal pyramidal neurons. Mol Psychiatry. 2019;24:588–600.
Prini P, Penna F, Sciuccati E, Alberio T, Rubino T. Chronic delta(8)-THC exposure differently affects histone modifications in the adolescent and adult rat brain. Int J Mol Sci. 2017;18:2094.
Orihuel J, Capellan R, Roura-Martinez D, Ucha M, Ambrosio E, Higuera-Matas A. Delta 9-tetrahydrocannabinol during adolescence reprograms the nucleus accumbens transcriptome, affecting reward processing, impulsivity, and specific aspects of cocaine addiction-like behavior in a sex-dependent manner. Int J Neuropsychopharmacol. 2021;24:920–33.
Weed PF, Filipeanu CM, Ketchum MJ, Winsauer PJ. Chronic delta9-tetrahydrocannabinol during adolescence differentially modulates striatal CB1 receptor expression and the acute and chronic effects on learning in adult rats. J Pharmacol Exp Ther. 2016;356:20–31.
Ballinger MD, Saito A, Abazyan B, Taniguchi Y, Huang CH, Ito K, et al. Adolescent Cannabis exposure interacts with mutant DISC1 to produce impaired adult emotional memory. Neurobiol Dis. 2015;82:176–84.
Rubino T, Vigano D, Realini N, Guidali C, Braida D, Capurro V, et al. Chronic delta 9-tetrahydrocannabinol during adolescence provokes sex-dependent changes in the emotional profile in adult rats: behavioral and biochemical correlates. Neuropsychopharmacology. 2008;33:2760–71.
Zamberletti E, Gabaglio M, Prini P, Rubino T, Parolaro D. Cortical neuroinflammation contributes to long-term cognitive dysfunctions following adolescent delta-9-tetrahydrocannabinol treatment in female rats. Eur Neuropsychopharmacol. 2015;25:2404–15.
Beggiato S, Ieraci A, Zuccarini M, Di Iorio P, Schwarcz R, Ferraro L. Alterations in rat prefrontal cortex kynurenic acid levels are involved in the enduring cognitive dysfunctions induced by tetrahydrocannabinol exposure during the adolescence. Front Psychiatry. 2022;13:996406.
Patrick ME, Fleming CB, Fairlie AM, Lee CM. Cross-fading motives for simultaneous alcohol and marijuana use: associations with young adults’ use and consequences across days. Drug Alcohol Depend. 2020;213:108077.
Wade NE, Wallace AL, Huestis MA, Lisdahl KM, Sullivan RM, Tapert SF. Cannabis use and neurocognitive performance at 13-14 years-old: optimizing assessment with hair toxicology in the adolescent brain cognitive development (ABCD) study. Addict Behav. 2024;150:107930.
Ehrenreich H, Rinn T, Kunert HJ, Moeller MR, Poser W, Schilling L, et al. Specific attentional dysfunction in adults following early start of Cannabis use. Psychopharmacology. 1999;142:295–301.
Duperrouzel JC, Hawes SW, Lopez-Quintero C, Pacheco-Colon I, Coxe S, Hayes T, et al. Adolescent Cannabis use and its associations with decision-making and episodic memory: preliminary results from a longitudinal study. Neuropsychology. 2019;33:701–10.
Lawn W, Trinci K, Mokrysz C, Borissova A, Ofori S, Petrilli K, et al. The acute effects of Cannabis with and without cannabidiol in adults and adolescents: a randomised, double-blind, placebo-controlled, crossover experiment. Addiction. 2023;118:1282–94.
Hanson KL, Winward JL, Schweinsburg AD, Medina KL, Brown SA, Tapert SF. Longitudinal study of cognition among adolescent marijuana users over three weeks of abstinence. Addict Behav. 2010;35:970–6.
Ertl N, Freeman TP, Mokrysz C, Ofori S, Borissova A, Petrilli K, et al. Acute effects of different types of Cannabis on young adult and adolescent resting-state brain networks. Neuropsychopharmacology. 2024;49:1640–51.
Mizrahi R, Watts JJ, Tseng KY. Mechanisms contributing to cognitive deficits in Cannabis users. Neuropharmacology. 2017;124:84–8.
Murray CH, Huang Z, Lee R, de Wit H. Adolescents are more sensitive than adults to acute behavioral and cognitive effects of THC. Neuropsychopharmacology. 2022;47:1331–8.
Mokrysz C, Freeman TP, Korkki S, Griffiths K, Curran HV. Are adolescents more vulnerable to the harmful effects of Cannabis than adults? a placebo-controlled study in human males. Transl Psychiatry. 2016;6:e961.
Kanayama G, Rogowska J, Pope HG, Gruber SA, Yurgelun-Todd DA. Spatial working memory in heavy Cannabis users: a functional magnetic resonance imaging study. Psychopharmacology. 2004;176:239–47.
Meier MH, Caspi A, Ambler A, Harrington H, Houts R, Keefe RS, et al. Persistent Cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci USA. 2012;109:E2657–64.
Schweinsburg AD, Schweinsburg BC, Medina KL, McQueeny T, Brown SA, Tapert SF. The influence of recency of use on fMRI response during spatial working memory in adolescent marijuana users. J Psychoact Drugs. 2010;42:401–12.
Solowij N, Stephens RS, Roffman RA, Babor T, Kadden R, Miller M, et al. Cognitive functioning of long-term heavy Cannabis users seeking treatment. Jama. 2002;287:1123–31.
Ceccarini J, Kuepper R, Kemels D, van Os J, Henquet C, Van Laere K. [18F]MK-9470 PET measurement of cannabinoid CB1 receptor availability in chronic Cannabis users. Addict Biol. 2015;20:357–67.
Hirvonen J, Goodwin RS, Li CT, Terry GE, Zoghbi SS, Morse C, et al. Reversible and regionally selective downregulation of brain cannabinoid CB1 receptors in chronic daily Cannabis smokers. Mol Psychiatry. 2012;17:642–9.
D’Souza DC, Cortes-Briones JA, Ranganathan M, Thurnauer H, Creatura G, Surti T, et al. Rapid changes in CB1 receptor availability in Cannabis dependent males after abstinence from Cannabis. Biol Psychiatry Cogn Neurosci Neuroimaging. 2016;1:60–7.
Pope HG Jr., Gruber AJ, Hudson JI, Huestis MA, Yurgelun-Todd D. Neuropsychological performance in long-term Cannabis users. Arch Gen Psychiatry. 2001;58:909–15.
Schreiner AM, Dunn ME. Residual effects of Cannabis use on neurocognitive performance after prolonged abstinence: a meta-analysis. Exp Clin Psychopharmacol. 2012;20:420–9.
Fontes MA, Bolla KI, Cunha PJ, Almeida PP, Jungerman F, Laranjeira RR, et al. Cannabis use before age 15 and subsequent executive functioning. Br J Psychiatry. 2011;198:442–7.
O’Shea M, Singh ME, McGregor IS, Mallet PE. Chronic cannabinoid exposure produces lasting memory impairment and increased anxiety in adolescent but not adult rats. J Psychopharmacol. 2004;18:502–8.
Schneider M, Koch M. Chronic pubertal, but not adult chronic cannabinoid treatment impairs sensorimotor gating, recognition memory, and the performance in a progressive ratio task in adult rats. Neuropsychopharmacology. 2003;28:1760–9.
Wiley JL, Evans RL, Grainger DB, Nicholson KL. Age-dependent differences in sensitivity and sensitization to cannabinoids and ‘club drugs’ in male adolescent and adult rats. Addict Biol. 2008;13:277–86.
Wiley JL, M O’Connell M, Tokarz ME, Wright MJ Jr. Pharmacological effects of acute and repeated administration of delta(9)-tetrahydrocannabinol in adolescent and adult rats. J Pharmacol Exp Ther. 2007;320:1097–105.
Gomes FV, Issy AC, Ferreira FR, Viveros MP, Del Bel EA, Guimaraes FS. Cannabidiol attenuates sensorimotor gating disruption and molecular changes induced by chronic antagonism of NMDA receptors in mice. Int J Neuropsychopharmacol. 2014;18:pyu041.
Jacobs-Brichford E, Manson KF, Roitman JD. Effects of chronic cannabinoid exposure during adolescence on reward preference and mPFC activation in adulthood. Physiol Behav. 2019;199:395–404.
Cha YM, White AM, Kuhn CM, Wilson WA, Swartzwelder HS. Differential effects of delta9-THC on learning in adolescent and adult rats. Pharmacol Biochem Behav. 2006;83:448–55.
Hashimotodani Y, Ohno-Shosaku T, Kano M. Presynaptic monoacylglycerol lipase activity determines basal endocannabinoid tone and terminates retrograde endocannabinoid signaling in the hippocampus. J Neurosci. 2007;27:1211–9.
Kim J, Alger BE. Reduction in endocannabinoid tone is a homeostatic mechanism for specific inhibitory synapses. Nat Neurosci. 2010;13:592–600.
Marcus DJ, Bedse G, Gaulden AD, Ryan JD, Kondev V, Winters ND, et al. Endocannabinoid signaling collapse mediates stress-induced amygdalo-cortical strengthening. Neuron. 2020;105:1062–76.e1066.
Neu A, Foldy C, Soltesz I. Postsynaptic origin of CB1-dependent tonic inhibition of GABA release at cholecystokinin-positive basket cell to pyramidal cell synapses in the CA1 region of the rat hippocampus. J Physiol. 2007;578:233–47.
Petrie GN, Balsevich G, Fuzesi T, Aukema RJ, Driever WPF, van der Stelt M, et al. Disruption of tonic endocannabinoid signalling triggers cellular, behavioural and neuroendocrine responses consistent with a stress response. Br J Pharmacol. 2023;180:3146–59.
Tanimura A, Yamazaki M, Hashimotodani Y, Uchigashima M, Kawata S, Abe M, et al. The endocannabinoid 2-arachidonoylglycerol produced by diacylglycerol lipase alpha mediates retrograde suppression of synaptic transmission. Neuron. 2010;65:320–7.
Barti B, Dudok B, Kenesei K, Zoldi M, Miczan V, Balla GY, et al. Presynaptic nanoscale components of retrograde synaptic signaling. Sci Adv. 2024;10:eado0077.
Bohme MA, McCarthy AW, Grasskamp AT, Beuschel CB, Goel P, Jusyte M, et al. Rapid active zone remodeling consolidates presynaptic potentiation. Nat Commun. 2019;10:1085.
Church E, Hamid E, Zurawski Z, Potcoava M, Flores-Barrera E, Caballero A, et al. Synaptic integration of subquantal neurotransmission by colocalized G protein-coupled receptors in presynaptic terminals. J Neurosci. 2022;42:980–1000.
Hruska M, Henderson N, Le Marchand SJ, Jafri H, Dalva MB. Synaptic nanomodules underlie the organization and plasticity of spine synapses. Nat Neurosci. 2018;21:671–82.
Newman ZL, Bakshinskaya D, Schultz R, Kenny SJ, Moon S, Aghi K, et al. Determinants of synapse diversity revealed by super-resolution quantal transmission and active zone imaging. Nat Commun. 2022;13:229.
Caballero A, Flores-Barrera E, Cass DK, Tseng KY. Differential regulation of parvalbumin and calretinin interneurons in the prefrontal cortex during adolescence. Brain Struct Funct. 2014;219:395–406.
Tseng KY, O’Donnell P. Dopamine modulation of prefrontal cortical interneurons changes during adolescence. Cereb Cortex. 2007;17:1235–40.
Tseng KY, O’Donnell P. D2 dopamine receptors recruit a GABA component for their attenuation of excitatory synaptic transmission in the adult rat prefrontal cortex. Synapse. 2007;61:843–50.
Urs NM, Gee SM, Pack TF, McCorvy JD, Evron T, Snyder JC, et al. Distinct cortical and striatal actions of a beta-arrestin-biased dopamine D2 receptor ligand reveal unique antipsychotic-like properties. Proc Natl Acad Sci USA. 2016;113:E8178–E8186.